In Part 1, Part 2, and Part 3 of this 4-part series, we covered the CKM framework (the heart–kidney–metabolic connection), cardiovascular risk assessment, what markers to measure, and lifestyle. This fourth and final part (#71) covers the medical side: when medication is needed, what the options are, and who qualifies for treatment regardless of their risk score.
The calcium scan: Seeing inside your arteries
Before we get to medication, there’s a test worth knowing about, one that often settles the question of whether medication is needed at all.
A Coronary Artery Calcium (CAC) scan is a low-radiation CT scan that detects calcium deposits in the walls of the arteries supplying blood to your heart. Calcium is a sign of plaque. The result of the scan is a single number: your CAC score.
Risk calculators give you a statistical estimate based on population data. A CAC scan gives you direct evidence of what is actually happening inside your arteries. It is particularly useful when your calculated risk is borderline because it moves the conversation from probability to physical evidence. The 2026 ACC/AHA (American College of Cardiology / American Heart Association) guidelines recommend considering it for men at or above age 40 and women at or above age 45 when treatment decisions are uncertain.
Here is how to interpret the CAC score:
- CAC = 0: No detectable calcium. Near-term risk is low, which is reassuring, but not a free pass, especially if you are younger or carry strong genetic risk like elevated Lp(a). Focus on lifestyle; medication can usually wait.
- CAC 1–99: Early plaque. A statin is reasonable; Aim for LDL below 100 mg/dL.
- CAC 100–299: Meaningful plaque. Statin recommended; Aim for LDL below 70 mg/dL, with at least a 50% reduction from your starting level (whichever is lower).
- CAC ≥ 300: High plaque burden. Aim for LDL below 55 mg/dL. Treat as you would established heart disease.
- CAC ≥ 1000: Very high plaque burden. Treat as established heart disease with aggressive therapy.
One important nuance: the same CAC score means very different things at different ages. My CAC score of 266 at 43 meant I was in the top ~2% for plaque for my age; if that were my score at 73, I would be closer to the 50th percentile. That is why percentiles, or how your score compares to others of the same age and sex, matter just as much as the number itself, and should always be read alongside the absolute score.
A note for south Asians: If your CAC score comes back anything other than zero, it is worth discussing a CT Coronary Angiography with your doctor. South Asians develop heart disease younger, more severely, and at lower cholesterol levels than Western risk calculators predict. A non-zero CAC score in a South Asian patient is a more significant signal than it might appear on its own. A CT Coronary Angiography provides a detailed picture of the arteries including soft plaque that the calcium scan cannot detect, and gives your doctor a much fuller view before deciding on treatment intensity.
Lipid targets: what the ACC/AHA says
The 2026 guidelines introduce ApoB goals alongside LDL targets. ApoB measures the number of cholesterol-carrying particles rather than just the cholesterol inside them, which is a more precise gauge of actual risk, especially for South Asians, people with diabetes, or anyone with elevated triglycerides where LDL can look deceptively normal. Once your LDL is at or near goal, checking ApoB tells you whether too many cholesterol-carrying particles still remain. ApoB targets follow the same risk-based thresholds as LDL: below 90 for intermediate, below 70 for high, and below 55 mg/dL for very high risk.
Lipid targets: What the Lipid Association of India (LAI) says
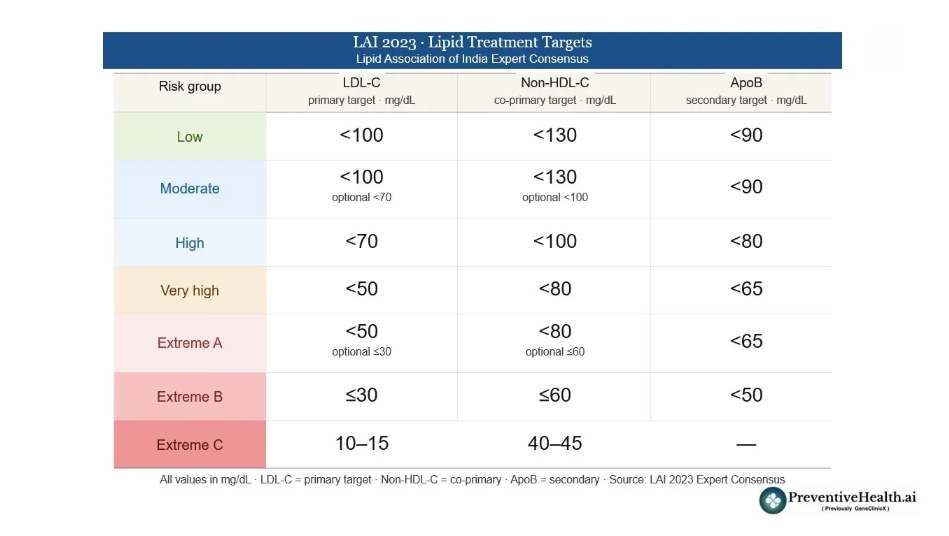
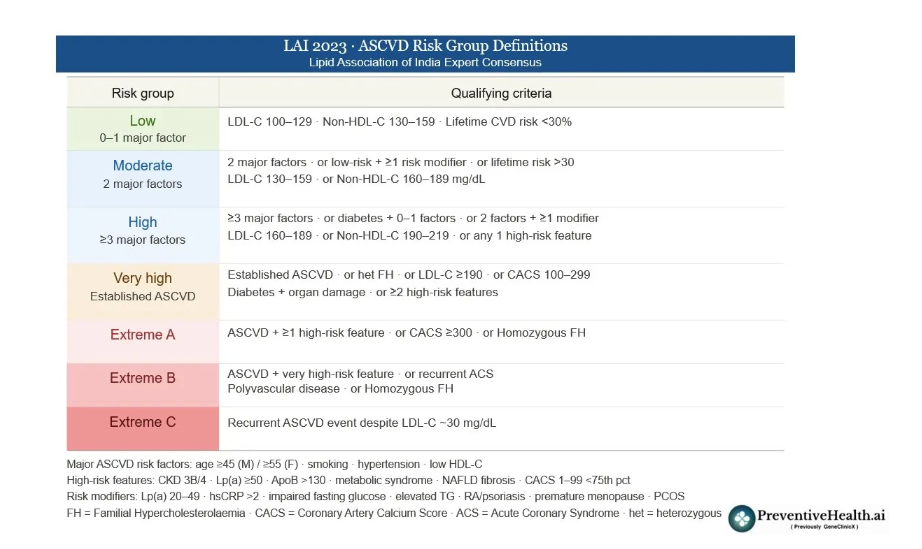
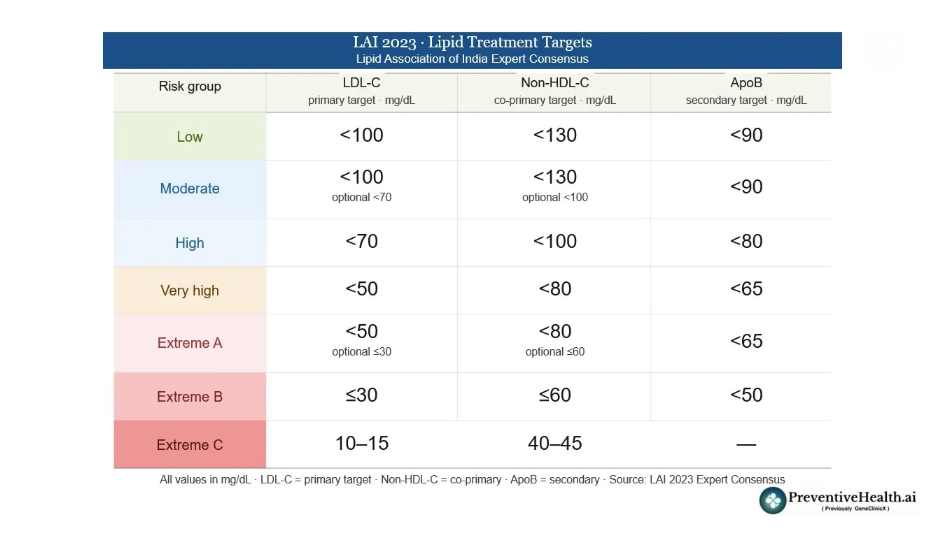
The Lipid Association of India (LAI) is India’s leading specialist body on lipid management. Its guidelines matter because Western-derived risk models were built on Western populations, and they can systematically underestimate risk in Indians, who develop heart disease roughly a decade earlier and with a more aggressive profile. Here is how the two sets of guidelines compare.
The most fundamental difference is their time horizon. The LAI places greater emphasis on lifetime risk and earlier intervention, whereas the ACC/AHA anchors decisions primarily on 10-year risk, with lifetime risk as a secondary lens.
On treatment targets, both recommend LDL below 70 mg/dL for high-risk patients. However, the LAI pushes harder for very high-risk individuals, recommending below 50 mg/dL versus the ACC/AHA’s below 55 mg/dL. The LAI also introduces three escalating extreme-risk categories with targets as low as 10–15 mg/dL, territory the ACC/AHA framework does not formally address.
Both guidelines use CAC scoring as a risk stratifier. The difference is in philosophy: the ACC/AHA uses CAC as a formal decision gate; scan first, then decide. while the LAI is more inclined to begin treatment earlier without necessarily waiting for imaging confirmation, given the earlier disease onset in South Asians.
Finally, the LAI gives Non-HDL cholesterol equal importance alongside LDL. This reflects the fact that South Asians tend to have higher triglycerides, which reflect more cholesterol-carrying particles beyond LDL; captured by non-HDL but not fully by LDL alone.. The ACC/AHA includes Non-HDL-C as a threshold but gives it less formal prominence.
Medications: The full ladder
If lifestyle is the foundation, medication is what you add when the foundation alone is not enough. The 2026 guidelines lay out a clear step-by-step approach.
Statins remain the starting point for most people. They reduce cholesterol production in the liver and are the most thoroughly studied, cost-effective option available. Around one in ten people experience muscle discomfort at full doses, so switching to a different statin or adjusting the dose usually resolves it. Statins modestly raise the risk of type 2 diabetes in people already predisposed, but the reduction in cardiovascular risk is significantly larger than the small increase in diabetes risk. If diabetes does develop, the guidance is to continue the statin and focus harder on lifestyle. My HbA1c went up from 5.8 to 6.1% when I started taking statins but it is now down to 5.6%.
Decades of clinical trials involving hundreds of thousands of patients have shown that statins not only lower LDL but directly reduce heart attacks, strokes, and cardiovascular deaths. The other medications on this list are newer, and while they lower LDL effectively, most of them do not yet have the same depth of evidence showing they reduce actual cardiovascular events. Statins are also inexpensive, widely available, and taken as a single daily tablet.
Can you stop statins once you start?
This is one of the most common questions I hear, and the honest answer is: stopping is not straightforward. Statins work by continuously suppressing cholesterol production in the liver. When you stop, LDL typically returns to its pre-treatment level within weeks — and with it, the cardiovascular risk you were managing. The protection is not banked; it requires the drug to be present. There are situations where a doctor might review the need, after age 75, for example, when the calculus becomes more individual. But in most cases, once statins are started for a meaningful cardiovascular indication, they are intended to be long-term. Think of them less like a course of antibiotics and more like blood pressure medication: the benefit lasts as long as the treatment does.
When statins are not enough or not tolerated:
- Ezetimibe reduces intestinal cholesterol absorption and lowers LDL by a further 15–20%.
- Bempedoic acid is an oral tablet, a useful alternative for people who cannot tolerate statins.
- PCSK9 inhibitors are injections given every two to four weeks that can cut LDL by 50–60% on top of a statin. They also modestly lower Lp(a), the inherited cardiovascular risk particle that diet and exercise cannot move. This can be quite expensive unless covered by insurance.
- Inclisiran is a newer RNA-based injection given just twice a year with similar cholesterol-lowering effectiveness.
For elevated triglycerides: If lifestyle changes are not enough, a prescription-strength omega-3 fatty acid has strong evidence for reducing cardiovascular events in high-risk patients whose triglycerides remain elevated on a statin. Statins remain the foundation even here; the omega-3 is added on top.
Who gets treatment regardless of their risk score
For most people, treatment decisions depend on calculated risk. But some groups are treated regardless:
- Anyone who has already had a heart attack, stroke, or other cardiovascular event
- Adults aged 40–75 with type 2 diabetes
- Chronic kidney disease (Stage 3 or higher)
- People living with HIV
- LDL at or above 190 mg/dL
- Familial hypercholesterolaemia: inherited high cholesterol, affecting roughly 1 in 250 people, most of whom do not know they have it
After age 75, decisions become more individualised, based on overall health and personal preferences.
If you have children: Don’t wait for symptoms
Guidelines recommend cholesterol screening for all children between ages 9–11, and again between 17–21. If you have high cholesterol or a family history of early heart disease, testing may start earlier. If a problem is found in one family member, testing close relatives catches risk early, before damage accumulates.
The bigger picture
What I find most encouraging about these guidelines is the shift in philosophy. We are no longer treating individual numbers in isolation. The CKM framework makes it clear that heart, kidney, and metabolic health are deeply interconnected. Risk is no longer just about LDL, and treatment is no longer reactive.
We now have better tools to measure risk, better ways to personalise decisions, and clearer pathways to act early. But the fundamentals have not changed. Consistent habits, better muscle mass, improved insulin sensitivity, and sustained lifestyle changes still drive the biggest impact.
That is it for this four-part series. I hope it has been useful and, as always, I would love to hear your thoughts and experiences in the comments.
Nickhil Jakatdar is the CEO of GenePath Diagnostics and the founder of PreventiveHealth.ai, working to make advanced diagnostics and credible health guidance more accessible, affordable, and actionable. A PhD in EECS from UC Berkeley and the youngest recipient of the UC Berkeley Distinguished Alumnus Award, he has founded and led multiple tech companies. He holds 60 patents, and serves on several academic, healthcare, and innovation boards. To follow his thinking on preventive health, technology, and systems that scale expertise, join his private WhatsApp community and subscribe to his Substack.
Disclaimer
Views expressed above are the author’s own.
END OF ARTICLE





